SalivaDirectTM is unique in 4 ways:
Saliva-based
You don’t have to stick an uncomfortable swab up your nose, which will increase testing compliance. This also protects healthcare workers from potential exposure to sick patients and works around worldwide swab shortages. We also found that expensive preservatives are not needed to test saliva, which lowers the costs.
Extraction-free
Nucleic acid extraction is time-consuming and expensive, and there have been worldwide shortages of the supplies needed to do it. For SalivaDirect™, Yale School of Public Health has worked out how to skip this step, making it more accessible.
Accurate
The SalivaDirect method reflects 100% Clinical Specificity and 94% Clinical Sensitivity.
Flexible
While many labs have similar equipment, they may be from a different company, and all of these need to be authorized by the FDA. We are aiming to validate our method with as many different variations of equipment and reagents as possible to allow labs to work with what they have and to prevent shortages. If your equipment isn’t on our list, let us know and we will work with you.
What is SalivaDirectTM ?
SalivaDirect™ was developed by the Yale School of Public Health to efficiently test for SARS-CoV-2. This requires a high complexity CLIA certified laboratory, such as Access Medical Laboratories to perform the testing. While the difficult RNA-extraction process has been removed, this test is still considered complex, as it requires PCR instrumentation. SalivaDirect also protects healthcare workers from potential exposure to sick patients, and works around worldwide swab shortages.


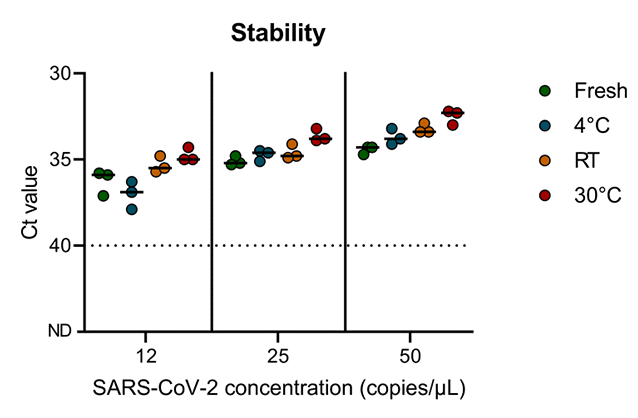
Stability
Stability of spiked-in saliva samples used for the limit of detection experiment was determined by placing 3 different concentrations (12, 25, and 50 copies/µL) for 7 days under different temperature regimes (4°C, room temperature, and 30°C). Results were compared to results obtained in the limit of detection experiment (fresh). Samples were treated with ThermoFisher Scientific proteinase K and tested with the ThermoFisher TaqPath RT-qPCR kit on the Bio-Rad CFX96. Overall, samples were stable under each condition for at least 7 days.
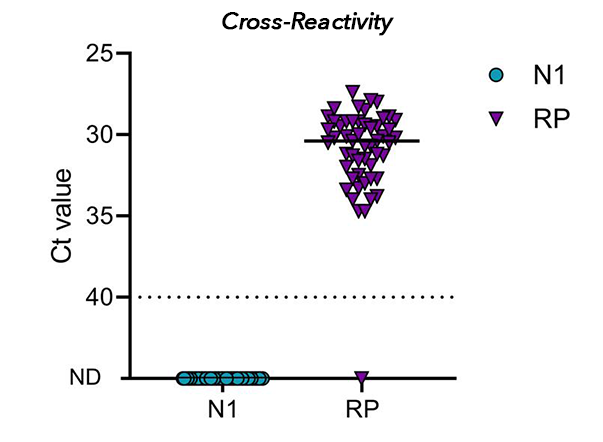
Cross-Reactivity
addition to cross-reactivity tests performed by the CDC, Yale has tested SalivaDirect™ on 52 saliva specimens collected from adults during the 2018/2019 and 2019/2020 (pre-COVID19) autumn/winter influenza seasons and detected 51 negative, and 1 invalid results.

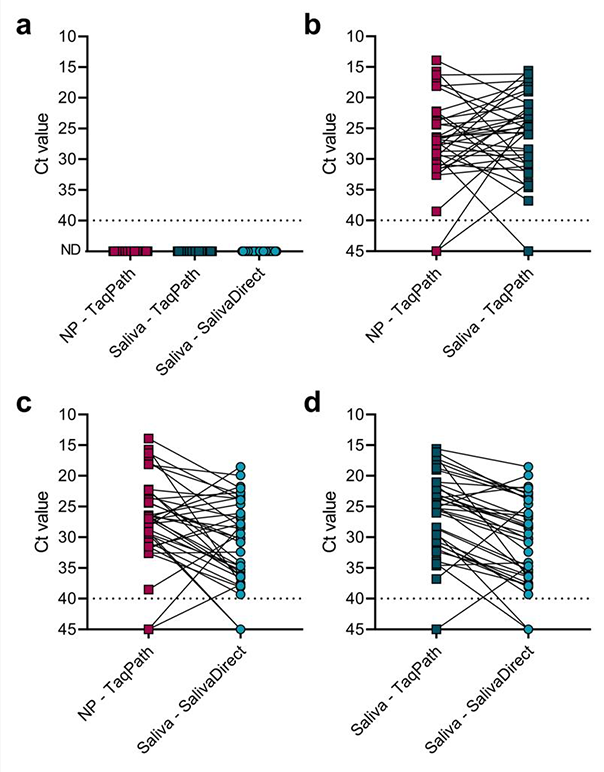
Clinical Validation
Performance of SalivaDirect™ was compared to the authorized ThermoFisher Scientific TaqPath RT-PCR COVID-19 kit by testing 37 paired positive and 30 paired negative nasopharyngeal (NP) and saliva samples. Nasopharyngeal swabs and saliva were collected from inpatients and healthcare workers in the Yale-New Haven Hospital. Saliva was collected in sterile urine cups or 5 mL tubes without addition of any preservatives. The ThermoFisher Scientific TaqPath COVID-19 combo kit combines RNA extraction using the MagMax Viral/Pathogen Nucleic Acid Isolation Kit with a multiplex RT-PCR diagnostic assay targeting 3 regions of the SARS-CoV-2 genome (N, S, and ORF1ab). For SalivaDirect™ we used the ThermoFisher Scientific proteinase K, ThermoFisher Scientific TaqPath RT-PCR kit, and Bio-Rad CFX96 instrument.
Yale University selected the positive and negative pairs based on preliminary results of our modified research protocol based on the US CDC primer-probe sets. Out of the 37 nasopharyngeal swabs that were tested with the TaqPath COVID-19 kit, three specimens tested negative (Ct value cut-off of 37). However, earlier results with the modified CDC assay indicated a (weakly) positive signal, and the paired saliva specimen tested positive with both SalivaDirect™ and the TaqPath COVID-19 kit. Two saliva specimens tested negative for SalivaDirect™ while the NP swab and saliva tested positive or inconclusive with the TaqPath COVID-19 kit.
All negative NP and saliva specimens tested negative with both the TaqPath COVID-19 kit and SalivaDirect™.