We didn’t invent the lab test,
We simply perfected it.
QFTB Gold +
QuantiFERON - TB Gold Plus
What is being tested in our QuantiFERON-TB Gold Plus?
What is QuantiFERON-TB Gold Plus?
By measuring a patient’s immune response to certain TB antigens, QuantiFERON-TB Gold Plus (QFT-Plus) aids in the diagnosis of mycobacterium tuberculosis infection. The panel detects responses to infections from the Mycobacterium tuberculosis (M.tb) complex. This includes some non-tuberculous bacteria, such as M Kansasii, M szulgai, and M marinum. The Gold Plus panel is approved by the FDA and is a forerunner in efficient and comprehensive tuberculosis testing.
The test is an interferon-gamma (IFN-γ) enzyme-linked immunosorbent assay (ELISA), a widely-used type of interferon-gamma release assay (IGRA). A combination of peptide antigens that stimulate secretions from the M tuberculosis bacterial complex, including proteins ESAT-6 and CFP10, are used to stimulate lymphocyte responses in heparinized whole blood. T cell responses to these peptides can indicate the presence or absence of M.tb.
What is measured in the QuantiFERON-TB Gold Plus?
QFT-Plus tests report on the cell-mediated immune responses of lymphocytes such as CD4 and CD8 T cells, both of which are recruited at the onset of M.tb infection. Patients that are infected with M.tb complex agents will respond to the peptide antigens through the secretion of IFN-γ. Specifically, CD4 T cells respond to TB1 antigens, and CD8 T Cells to TB2 antigens. These two T cells are largely responsible for IFN-γ secretions as a defense against TB peptide antigens. Therefore, by measuring IFN-γ levels in the plasma, QTF-Plus panels can detect m. tuberculosis infection already extant in the patient
CHEST PAIN WITH BREATHING
FATIGUE
FEVER
Chills
COUGH UP BLOOD OR MUCUS
How does testing contribute to patient care?
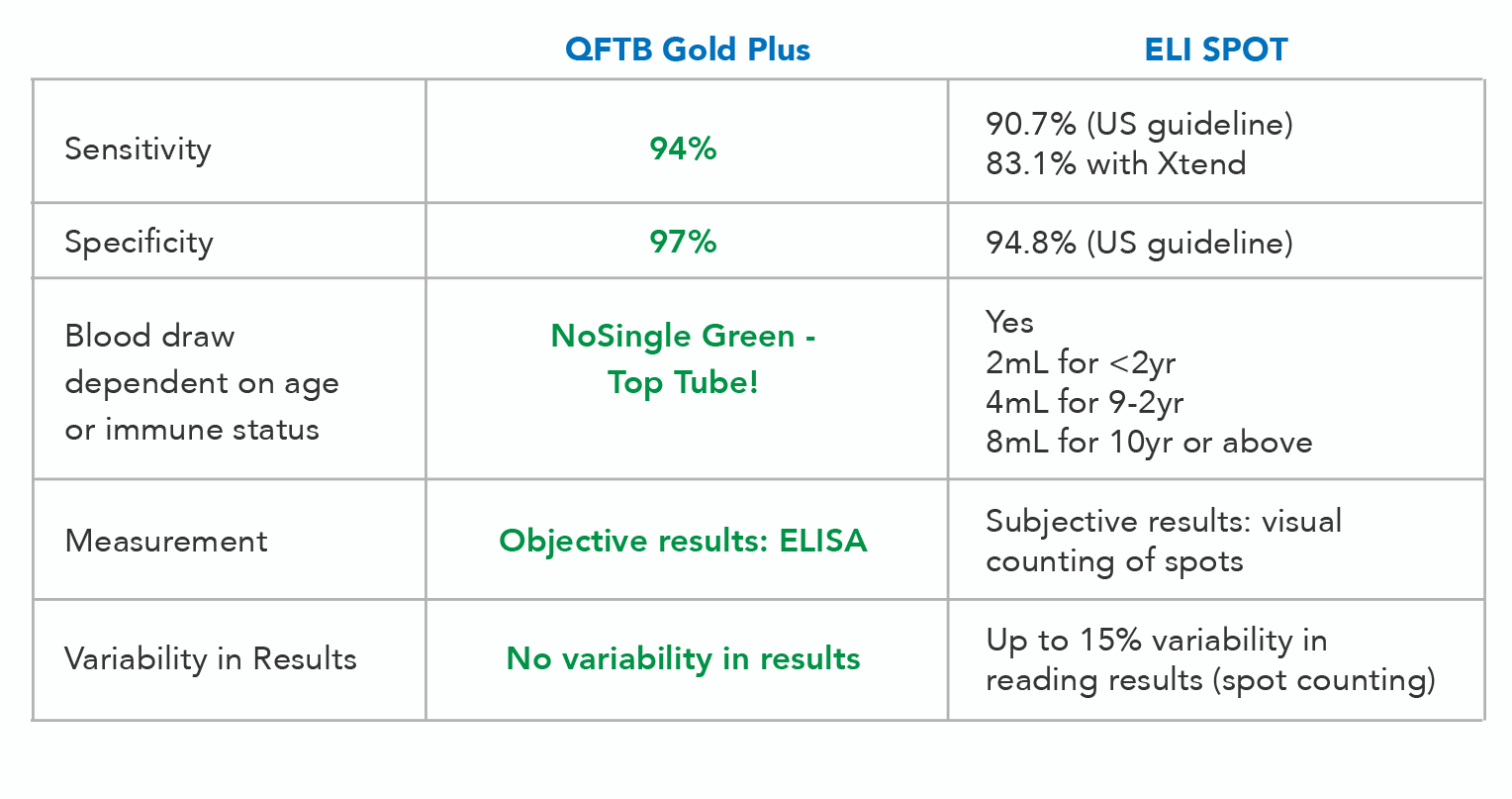
With additional diagnostic evaluations, such as radiography and risk assessment, a positive QFT-Plus result can confirm a TB diagnosis. The test has a 94% sensitivity level and a 97% specificity level. Fewer false positives and negatives mean faster diagnosis.
Vitally, QFT-Plus can detect both latent TB infections (LTBI) and active TB disease. As a highly infectious disease, early detection of TB as a non-infectious latent tuberculosis infection before it develops into active tuberculosis is invaluable.
What makes QuantiFERON-TB Gold Plus different from other TB testing?
Increased accuracy: QFT-Plus is more accurate than other forms of TB testing. False-positive results are reduced in QFT-Plus compared to TST. The chemiluminescence immunoassay used by Access Medical Labs has a higher level of specificity and sensitivity than even ELISA, making it the premier methodology for tuberculosis testing. Although indeterminate test results occasionally occur, due to high nil values or low IFN-γ in the mitogen tubes, the test’s objective measurements eliminate result variability.
The Gold Plus test is at the forefront of TB testing. Unlike prior iterations of the test, such as the Gold In-tube, Gold Plus tests for both CD4 and CD8 T cell reactions for the most comprehensive analysis of your patients’ immune responses.
Improved accessibility: Unlike TST, which requires a follow-up appointment to complete testing, QFT-Plus requires only a single patient visit, making it more accessible for both you and your patients. QFT-Plus doesn’t require you to centrifuge the blood samples. Blood is collected into a single lithium heparin blood collection tube and can be held at room temperature for up to 16 hours before incubation.
What is the testing process?
All blood samples must be collected at a doctor’s office, clinic, or hospital. Once your sample is received in our laboratory, our team of highly-trained lab technologists will analyze the sample and send the results to you and/or your healthcare provider within 24-48 hours. Find additional information or order a QuantiFERON-TB Gold Plus panel here.